|
|
Measurement of the Glass Transition Temperature Tg
Determination of the glass transition temperature, Tg, by dilatometry:
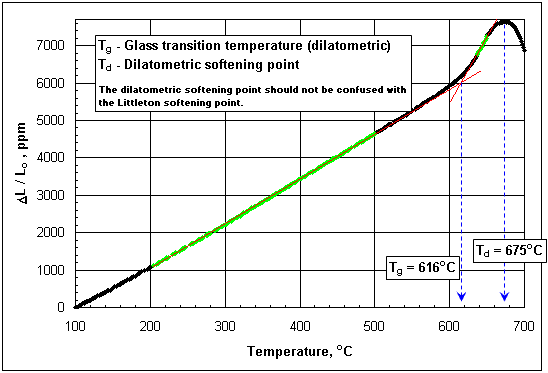
Figure 1: Determination of Tg by dilatometry (click image to enlarge). The linear sections below and above Tg are marked green; Tg is the temperature at the point of intersection of the corresponding red regression lines.
Tg may be derived from the thermal expansion curve as demonstrated in Figure 1. It is not clearly defined, however, how the slope changes and the green marked linear sections should be found. It is necessary to rely in a reasonable personal judgement.
Determination of the glass transition temperature by DTA/DSC:
For most Tg values in the literature the DTA/DSC technique is used. The following paragraphs are taken from a paper by Mazurin & Gankin (2007) about the subject:
Probably everybody agrees with the statement that Tg is one of the temperatures within a glass transition region where the metastable melt transforms into glass in the course of its cooling and where glass transforms into metastable melt in the course of heating. The exact value of Tg is determined by analyzing the temperature dependence of a property. To do this some scientists use cooling curves. However, the overwhelming majority of scientists prefer to use solely heating curves. Below we will consider the results of analyzing of only these kinds of heating curves.
Figure 2 shows three different points on the curve of temperature dependencies of specific heat that can be used to determine Tg. In more than 300 DTA/DSC studies of Tg published over a period of 2005-2006 the authors specified the way of Tg determinations only in 68 publications. 70% of these authors used point A, 20% point B and 10% point C. In about 250 papers (out of 300) the way of Tg determination was not specified. At the same time, as is seen from the caption to Figure 2, the difference between these three values of Tg is quite great. It seems obvious that to standardize Tg determination it is necessary to select the most popular way of processing DTA/DSC curves, namely, the intersection of two tangents at the start of the corresponding endotherm (point A).
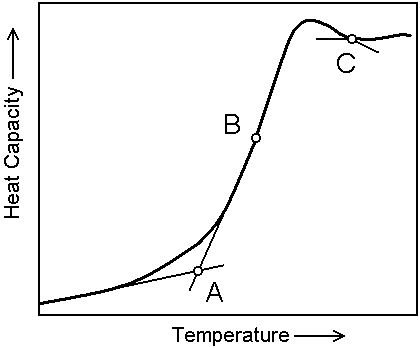
Figure 2: Various points in temperature dependencies of heat capacity used to determine Tg. The values of log(η(Tg), Poise) for these points in the case of commercial soda-lime-silica glasses (in the temperature range of 500- 580°C viscosity was calculated by the equation log(η(Tg), Poise) = -22.56 + 28200/T where T is the absolute temperature): A: 13.20; B: 12.36; C: 11.56. The calculations were done for heating rate of 10 K/min and for glass samples initially cooled from the temperatures well above Tg at the same rate.
It is well known that the values of Tg depend both on heating rates and rates of cooling of samples prior to measurements (Mazurin, 2007). The ratio of cooling and heating rates has also an influence on the shape of DTA/DSC curves, which in its turn influences the reproducibility of the results obtained. The optimum results are obtained in the cases when an initial cooling rate is equal to a heating rate (see, for example, Moynihan et al., 1974). There are many cases when samples used for measurements were preliminary annealed using a certain arbitrarily selected schedule. In these cases it is practically impossible to predict the result, except that the deeper the annealing, the lower the log(η(Tg)) value.
To obtain Tg values compatible with each other (i.e. the values that will allow for the joint use of data of numerous authors in order to find general composition dependencies in wide composition ranges) it is necessary to select a certain standard heating rate for all experiments. The analysis of the experimental data on Tg published in 2005 shows that 75% of investigators specifying heating rates for DTA/DSC measurements selected 10 K/min. It should be taken as a standard heating rate. Correspondingly, the rate of initial cooling should also be equal to 10 K/min. For dilatometric measurements of Tg, however, standard cooling/heating rates within the range of 3-5 K/min should be used. In this case, Tg values obtained by DTA/DSC and dilatometric methods will be approximately compatible with each other. This statement was substantiated in some details by Mazurin (2007).
One of the main obstacles to the efficient use of Tg data is the fact that in more than half of all papers containing Tg data it is impossible to find information required for a proper application of such data. As follows from Figure 2 and Mazurin (2007) for samples of the same glass a possible range of changes in log(η(Tg), Poise) can be found between ~14.0 (determination of Tg by version A for quenched samples heated at the rate of 3 K/min) and ~10.0 (determination of Tg by version C, for well annealed samples at the heating rate of 40 K/min).
If a scientist wants to publish Tg data that could be used efficiently by other scientists, it is necessary to follow several quite simple recommendations: (1) to use version A for determination of Tg (see Figure 2); (2) to select a heating rate equal to 10 K/min in DTA/DSC studies and 3-5 K/min in dilatometric studies; (3) to cool the samples used for measurements from a temperature well above Tg at the rate equal to a heating rate in the course of measurements; (4) to specify the above three features of the experiments in the a paper to be published.
Mazurin (2007)
O. V. Mazurin; Glass Physics and Chemistry, vol. 33, 2007, p 22.
Mazurin & Gankin (2007)
O. V. Mazurin, Yu. V. Gankin: "Glass transition temperature: problems of measurements and analysis of the existing data"; Proceedings, International Congress on Glass, July 1-6, 2007, Strasbourg, France.
Moynihan et al. (1974)
C. T. Moynihan, A. J. Easteal, J. Wilder, J. J. Tucker; J. Phys. Chem., vol. 78, 1974, p 2673.